 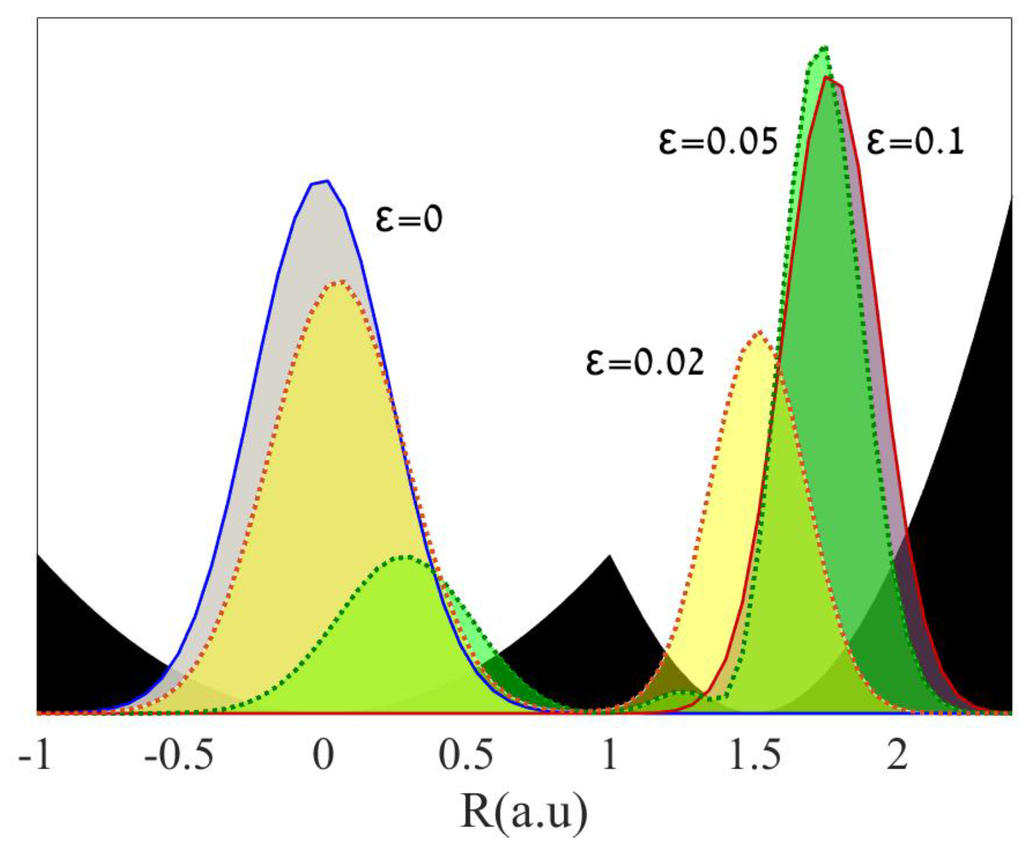
This means, of course, that the quotient q/ T cannot be a state function either, so we are unable to use it to get differences between reactants and products as we do with Manner in which a process is carried out. It turns out that we can generalize this to other processes as well, but there is a difficulty with using q because it is not a state function that is, its value is dependent on the pathway or You will recall that when a quantity of heat q flows from a warmer body to a cooler one, permitting the available thermal energy to spread into and populate more microstates, that the ratio q/ T measures 
Sharing of energy can be related to measurable thermodynamic properties of substances – that is, of reactants and products. Now we need to understand how the direction and extent of the spreading and We have explored how the tendency of thermal energy to disperse as widely as possible is what drives all spontaneous processes, including chemical reactions. Rudolf Clausius originated the concept as energy gone to waste in the early 1850s, and its definition went through a number of more precise definitions over the next 15 years. It is also widely misrepresented as a measure of disorder, as we discuss below. Absolute entropies of most common substances are tabulated, allowing us to calculate the entropy of a reaction in the same way we can calculate enthalpy of reaction from standard enthalpies of formation.Įntropy is one of the most fundamental concepts of physical science, with far-reaching consequences ranging from cosmology to chemistry. The second section discusses the meaning of entropy, and what disorder means on a microscopic level.Įntropy is a state function, which means we can apply Hess' Law to it. A reversible process can be modeled as a series of tiny steps, while an irreversible process must be modeled as a single large change. The first section explains the difference between reversible and irreversible processes. 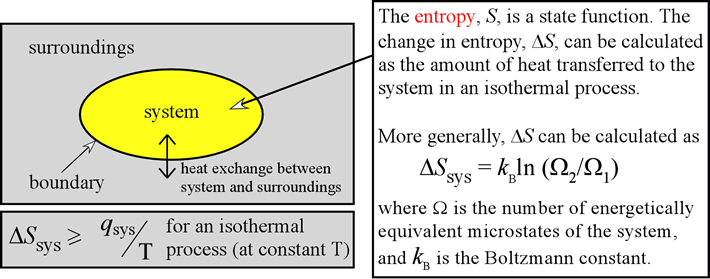
Because of this, the second law of thermodynamics explains why a perpetual motion machine can never exist. This is because some of the energy from your car engine is lost as heat. If you touch the hood of your car while the engine is running, the hood of the car will feel hot. One consequence of the second law of thermodynamics is that in any engine there will be some energy lost as heat that cannot be harnessed to do work. The second law of thermodynamics states that the entropy of the universe is always increasing. Entropy is a measure of the disorder of a system, measured in joules (J). If a “Pillet style” exists, it is in his ability to crystallize, within a project, the excitement of a proposition.The second law of thermodynamics involves a thermodynamic quantity we call entropy (S). Whether it be as design director for renowned fashion brands, or in longterm collaborations with first class furniture brands, his considered interpretations are a testo high-voltage chic, distinguished both by its precision and rigor. Architecture objects, furniture, artistic direction, his signature is invariably associated with the best furniture companies and with the finest luxury brands: Trussardi, Lacoste, Moët et Chandon, Veuve Clicquot, Lancome, Shiseido, l’Oréal, JC Jitrois, Catherine Malandrino, Lancel, Le Tannneur International… The scope and the variety of his projects share a common attitude, independent of scale. He has won international acclaim for the spectrum and quality of his creations. Pillet’s perfect command of sensuality and refinement has made him one of the rare French designers who have gained global recognition designing hotels, boutiques and directing artistic projects in Europe, USA, and Japan. Lucidity of expression and the search for simplicity are the key principles: in the work of Christophe Pillet the elegance is optimized. During his career he has published many articles on the most important magazines of the world. Pillet was elected as “Créateur de l’année 1994”. Since 1993, he manages his own design agency. He worked with Martine Bedin in Milano from 1986 to 1988 and cooperated with Philippe Starck, in Paris, from 1988 to 1993. 
Christophe Pillet graduated from the Decorative Arts School in Nice in 1985, Master of Domus Academy Milano, in 1986. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
